Acid Reflux To Counter Jacket Leakage
Table of Contents
HCl Synthesis Process Overview #
The synthesis of hydrogen chloride from chlorine and hydrogen is an exothermal reaction. For each mol of hydrogen chloride generated, \( 92.05 \ \text{kJ} \) of heat is released. The synthesis furnace is designed with a “3-in-1” graphite jacket to maximize heat recovery through by-product steam generation. The unit is divided into three primary sections: Combustion, Steam Co-generation, and Cooling.
The synthesis is represented by the following thermochemical equation:
\[ \text{H}_2(g) + \text{Cl}_2(g) \rightarrow 2\text{HCl}(g) \quad \Delta H = -184.1 \ \text{kJ/mol} \]
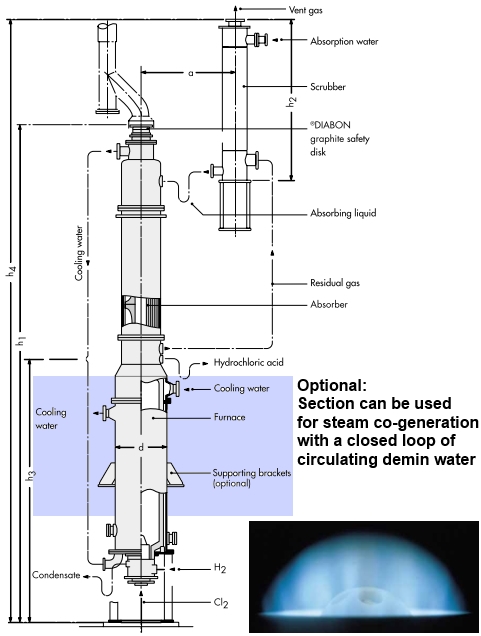
Feed and Ignition #
Hydrogen and chlorine are sourced from the electrolyser and pre-treatment sections. An orifice flowmeter normalizes the flow by accounting for gas pressure and temperature. The gases are then fed into the furnace burner at a controlled molar ratio \( 1.05:1 \) to \( 1.1:1 \). Burner material is typically silica glass or ceramic-based. An automatic electric igniter is used to initiate flame.
Combustion #
The resulting hydrogen chloride gas reaches temperatures between \( 2000 \) and \( 2500 \ ^\circ\text{C} \) as it flows from the combustion chamber.
Steam Generation #
Recirculating pure water absorbs the reaction heat of chlorine and hydrogen synthesis (generally 0.7 ton of by-product steam is generated per ton of 100% HCL, and 60% of the reaction heat is recycled). The steam can be conveyed to caustic soda plant for salt dissolving process or other processes, to save energy and reduce consumption.
Absorption #
It then enters an integral or separate isothermal co-current falling-film absorber. Here, the gas is absorbed in water or weak acid to produce hydrochloric acid at the desired concentration.
Leaking Graphite Block #
Source SMath file uploaded here